Immunization Developments on August 28, 2025
August 28, 2025
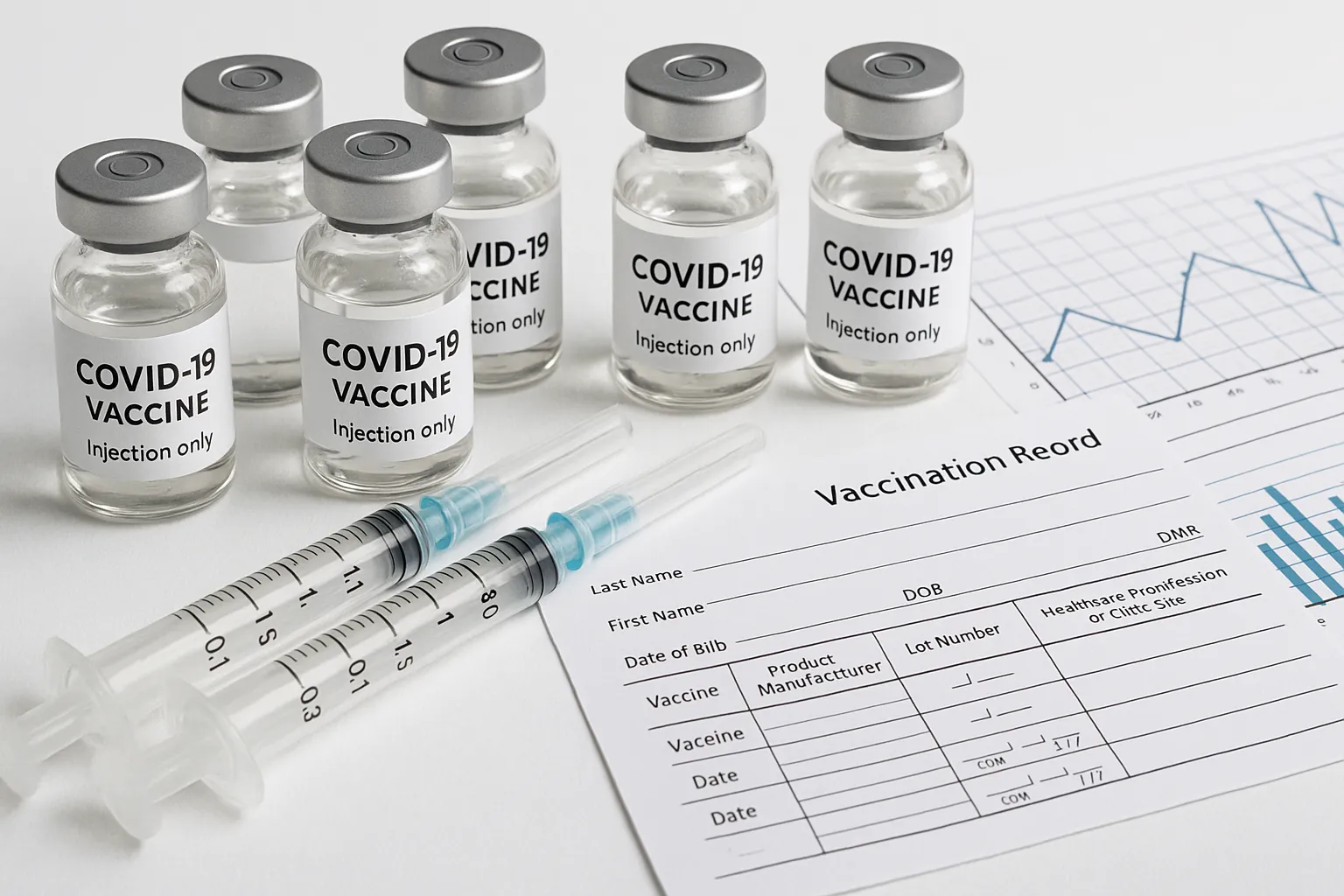
United States (Washington, D.C.) The U.S. Food and Drug Administration (FDA) this week issued new guidance narrowing eligibility for the updated 2025-26 COVID-19 vaccines. Under the revised authorization, only individuals aged 65 and older or those with underlying medical conditions may receive the booster shots without further review; healthy adults under 65 must first consult a healthcare provider before vaccination. The guidance, signed off by FDA leadership but still subject to endorsement by the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices, marks the most restrictive COVID-19 vaccination policy since the initial rollout in late 2020. Reaction has been swift: several CDC officials resigned in protest, and leading medical societies have issued conflicting recommendations in response to the changes.
Philippines (Manila) The Department of Agriculture announced approval and imminent rollout of Volvac B.E.S.T. AI + ND, the country’s first commercial vaccine against highly pathogenic avian influenza (H5N1) combined with protection against velogenic Newcastle disease. Developed by Boehringer Ingelheim, the recombinant vaccine will be administered by injection into poultry aged at least 10 days, with full immunity developing within two weeks. Officials hailed the move as a critical defense for poultry farmers and national food security, noting the vaccine’s dual action against two of the sector’s most devastating viral threats.
Global (Geneva) The World Health Organization (WHO) released its Multi-Country Mpox External Situation Report No. 57, warning that monkeypox (mpox) virus clades continue to circulate and pose a global health risk. Between July and August 2025, 47 countries across five WHO regions reported 3,924 confirmed cases and 30 deaths (CFR 0.8%). WHO Director-General Dr. Tedros Adhanom Ghebreyesus extended standing vaccination recommendations for high-risk groups and international travelers through August 20, 2026, to curb sustained community transmission.
Categories
Autos and vehicles Beauty and fashion Business and finance Climate Entertainment Food and drink Games Health Hobbies and leisure Jobs and education Law and government Other Politics Science Shopping Sports Technology Travel and transportationRecent Posts
Tags